
CPG, Food and Beverage, Cosmetic and Personal care: a blog dedicated to NPD issues and software
Product Information File (PIF) – All You Need to Know
Since the application of the new European Cosmetics Regulation 1223/2009 article 11 on cosmetic products sold in the European Union, industry professionals have to provide a Product Information File (...

Why use a PLM solution as a Nutrition label software?
Machines arrived in our lives in the 19th century. They became essential tools even if they caused some contradictory feelings like fear. For instance, they help us but sometimes they replace us. Tod...

Keeping Your Skin Care, Cosmetic and Fragrance Business Competitive With a PLM
Do you find yourself working against the clock to bring innovative and safe products to market? Are you staying ahead of new regulations and market trends? Remaining agile in the skincare, color co...

ERP and PLM: How Complete Integration Delivers the Best Results
Product lifecycle management (PLM) and enterprise resource planning (ERP) are well-known solutions for consumer goods companies. But, what exactly is the role of each one? And what benefits can your b...

6 Key Reasons Your Consumer Packaged Goods Business Needs a PLM System
Feeling the pressure to deliver more new products with a shorter time to market? All while streamlining your operations? With changes in consumer tastes and growing pressure to provide supply chain...

5 Signs You Need to Ditch Spreadsheets and Move to a PLM Solution
Spreadsheets, such as Microsoft Excel, are extensively used in the corporate and manufacturing world for maintaining contact information, making calculations and developing dashboards and comparison c...

4 Best Practices to Speed up your Food Product Development
Streamlining your product development process is essential to ensure that your deadlines are respected and your products hit the shelves on time. It is also important to create a smooth process as...

3 Tips To Better Manage Food Allergens
Food allergens are a real concern in the US. According to the FDA, about 2% of adults and 5% of infants and young children suffer from food allergies. Each year, it is estimated that approximately 30,...

Optimize Packaging Management with a PLM Software
Product packaging is a significant component of the new product development or renewal process. Brands use it as a way to create consumers’ demand, as it is the only way to inform and convince them ab...

Key features in food project management solutions
Project management is a fundamental issue for today’s businesses. It is increasingly common to hear “we want to work in project mode”, implying a willingness to plan, communicate and collaborate as be...

Carbon score & Agri-food: what are we talking about?
Feeding within the limits of our ecosystem is a key global issue as dynamics are intertwined. Consumers are becoming increasingly aware of environmental concerns, in addition to their social, nutritio...

The critical importance of multilingual data in CPG
As we discussed previously, global product management and development should not drag your international strategy. While we identify regulatory compliance management as the biggest business challenge...

The innovation cycle, a process to be managed throughout the product lifespan
The innovation cycle: a constantly changing concept The way innovation operates has changed rapidly over the last twenty years, pushed by technological advances. The very concept of innovation has al...

Formulation tool: the issues and requirements surrounding product design
Current issues in product formulation The nature and composition of products reaching the market nowadays has certainly changed a great deal. From a relatively straightforward offering in the past, w...

Food and Beverage Product LCA – Lifecycle Assessment
In these days of heightened concerns over environmental matters, product life-cycle assessment (LCA) plays a crucial role not only in businesses’ technology choices, but also in their long-term strate...

The puzzle of food & beverage and cosmetic regulatory intelligence
One of the central problems of the consumer-packaged goods industries is to ensure regulatory compliance of products marketed in one or more markets. In order to do this, companies need to be able to...

What constraints weigh in on CPG product development?
When taking part daily in New Product Development (NPD) projects, it may become difficult to take a moment and step back to look at the big picture again. This is what we offer you today. An overview-...

Eco design : The challenge of reducing fine particles emissions
As we have seen in a previous article, environmental protection is a critical issue in our society. If physical pollution, such as packaging, is a major problem, the impact of air pollution should not...

Cost, Time, Scope triangle – The case of PLM projects
The project management triangle, or Cost, Time, Scope triptych, is a basic, in both fundamental and simplistic meanings, concept of project management. It aims at illustrating the interdependence and...

How PLM can guide green manufacturing in CPG Industries
Energy is at the heart of our industrial society and it is no surprise that a majority comes from fossil energy (80% of the world's consumption is attributed to oil, gas, and coal). For example, in on...

What a PLM implementation project will require from your teams
PLM projects range from a very basic set of core features in order to begin a digitalization process, to a comprehensive and holistic new product development solution sustainably changing your whole p...

What are the main CPG software?
Digitalization induced major changes in the corporate world. Companies felt indeed the necessity to redefine their position and organization to meet the new habits created by new technologies. The cli...

Global product management should not drag your international strategy
Going international is not only a beautiful ambition (almost) every manufacturer aims for, it is also a highly strategical choice. Even though a company planning to start its global expansion does so...

Responsible consumption of water in product development
In a previous article, we explained why sustainability is important and presented 5 topics on which companies can still act. Today, we will explore the first one: water. It seems obvious but humankind...

“What is PLM software?’ breakdown – Part 2
1. What is PLM software’s most important concept to remember / grasp? The most important benefit of a PLM solution, and what all of its other features are based upon, is building a single version of...

“What is PLM software?” breakdown – Part 1
Answering the question “What is PLM software?” is not the matter of a single blog post. For a first-level presentation of PLM technology, we invite you to read our dedicated page. Here, we will try t...

The importance of tailoring your PLM system to your industry
A PLM (Product Lifecycle Management) system is a collaborative tool allowing to manage the entire product lifecycle of a manufactured product. It gathers in a unique and central repository all product...

Having the best PLM software is a matter of selection
To choose the best Product Lifecycle Management (PLM) software for your Consumer Packaged Goods (CPG) company, you should first have an idea of what PLM technology is, what benefits it provides, how i...

Cloud PLM or On-Premise PLM?
The comparative advantages of hosting your solution and data internally or externally have been widely discussed. However, we want to share our take on how this specifically affects your decisions whe...

The three pillars of sustainable product development
It is common knowledge that Earth’s resources are limited, and with larger populations and higher consumption habits, mankind is more and more challenged by coexisting with wildlife, pollution, and cl...

The product database – PLM basics
Product databases- or should we say, product information databases- are an unskippable item of any manufacturer’s digital landscape. That is obvious by now. But what is a product database? What tangib...

Major PLM project risks and how to mitigate them – Part 2
Once the PLM project has been properly set on track, it is important to stay alert to avoid it drifting away. There is considerable literature on project management and project management risks so we...

Major PLM project risks and how to mitigate them – Part 1
We intend in this 2-part blog post to give you a check-list of the main risks you face in any Product Lifecycle Management solution implementation project. This first part will cover "pre-project" con...

Cloud and SaaS adoption in Food and Beverage Industry
With the digitalization of mostly everything, companies have followed by reviewing their information systems and beginning their digital transformation. Since the early 2000s, cloud computing, or “re...

Can PLM and Blockchain complement each other?
Not only a health issue From 2004 through 2013, undeclared allergens were a leading cause of food recalls in the U.S., accounting for 27.4% of all recall events. Globally, barely 10% of food and beve...

Premiumization of CPG and the impact on product development
The global beauty industry registered a 5.5% growth in revenue in 2018 and is now valued at $532 billion. The global food and beverage industry is growing at around 5% a year and is expected to reach...

SMBs’ digitalization must also go through PLM adoption
If you are part of the 48% of Small and Medium Businesses (SMBs) currently planning to begin their digitalization journey or the 36% that are already on the way, we guess one of your biggest challenge...

Manage your product portfolio, then innovate!
As a manufacturer, you know the drill: legislation get stiffer, competition gets fiercer, consumers change their minds faster, etc. To face it, you may have put efforts in maximizing your innovation p...

How to make incorrect batch sheets
Determining the product’s consistency and conformity, the batch sheet counts as one of the most critical documents of the product lifecycle. Errors in the batch sheet may result in non-compliant prod...

Collaboration between FMCG suppliers and retailers as key to success
As consumers grow more demanding, retailers need to ensure products are available at the right time, through the right channel and at the right price. While it seems collaboration between retailers an...

Improving your new product development and reducing your time to market
It would be an understatement to say that there are many challenges associated with creating a product. Let alone dozens. There are just some things in today’s day and age that should be simpler, quic...

How to optimize change management process with PLM software?
Product change is a constant in the food industry and consumer packaged goods: New consumer needs and wants implies product formulas’ changes, Changes to suppliers and manufacturing processe...

PLM ERP – Why Do You Need to Combine Them?
PLM ERP are two major systems essential to develop and launch innovative and profitable products; therefore, it is important to understand their relationship. Indeed, ERP and PLM are complementary sof...

US: What are your labeling requirements for food products?
When it comes to prepacked food, its labeling can be as complex as either its pack development or its manufacturing process. Indeed, wherever it may be sold, prepacked food is subject to very strict r...

Best practices to avoid slowing down innovation processes
Whatever the food and beverage segment, a product is often changing. Change is the rule not the exception. Companies innovate constantly; even "best-sellers" need updates to remain profitable. Launch...

What is an ingredient statement? Zoom on the US and European requirements
Ingredient statement (or ingredients list) is a mandatory section for packed food labels in most international standards. Its aim is to inform the consumer about the food composition. In Europe, the r...

How to reduce time to market?
In food companies, products change quickly, as well as the number of competitors in the market. Innovation is important to develop and manage a coherent product portfolio that meets the ever changing...

How to cope with raw material prices volatility?
The cost of raw materials remains the top overall concern for food industries and manufacturers. Regarding extreme price volatility of raw materials it becomes a real challenge for food companies to r...
Regulatory trends of Brazilian and Chinese cosmetic markets
Exporting cosmetic products to China or Brazil is as profitable as it may be complex regarding their regulatory frameworks. In Paris, a few days ago, took place the annual COSMED meeting, dedicated t...

Worldwide regulatory framework for controls and inspections on cosmetic products
On March the 22nd, 2019 took place in Paris the COSMED annual international meeting regarding regulatory compliance management for cosmetic products. North American, Asian, and South American regulati...
Food Labeling: Food Companies Urged to Simplify Food Date Labels
The Consumer Goods Forum (CGF) – a network of 400 members across 70 countries – has urged food companies to simplify food date labels by 2020 in an effort to reduce consumers’ confusion related to “us...

How to adapt to food regulations such as INCO and FSMA?
All countries govern through a complex set of laws and regulations for the food that enters and leaves that particular country. These regulations result in demands which participants in the food chain...

PLM & ERP: complementary tools
ERP systems enable organizations to plan for orders and profitability by providing manufacturers with a real-time picture of their financial status. With an ERP system in place, companies can track or...

Case Study: How to optimize the cycle of innovation and production in the dairy sector?
Today, I decided to look at the product management in the dairy sector and to look at the best way to represent your benefits of a PLM solution with it being applied to this case study. Let’s take...

Safety Data Sheet: What you Need to Know
Safety Data Sheets (SDS), also known as Material Safety Datasheet (MSDS), are an essential component of the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). Created by the...

7 Steps to Optimize Cosmetic Product’s Formulation
Formulating a cosmetic product is a long and complex process as it needs to comply with marketing requirements – shape, color, function, texture, etc. but also with regulations enforced in the country...

How to Guarantee The Accuracy of Your Specifications ?
Product specification : Download our solution brief During a product development, it is important to ensure that the specifications are accurate, from receiving the raw materials, through formulati...

Cosmetic Products Compliance : 6 Tips to Better Manage Business Processes
Product compliance is one the main pain points when developing a new cosmetic. Regulations are evolving fast and it is getting more and more difficult to follow. With globalization, brands are commerc...

Food Software – Which Solution to Facilitate Your Product Development?
During a product development, Food and Beverage companies face various internal and external challenges. From shorter time-to-market, through a growing number of regulations and fast-evolving tech...

Challenges and best practices around food labeling compliance
Food labeling requirements: plenty of challenges Properly managing product labels is crucial to streamline a product development. However, there are many food labeling requirements to take into con...

Food Contact Materials
What Are Food Contact Materials? Food Contact Materials (FCMs) are materials that are already or are intended to be in contact with food and beverages. FCMs also includes materials that can reasonabl...

5 Tips to Better Manage your Cosmetic Product Specifications
Every new product comes with its specification that defines its identity and characteristics for both internal and external use. Often, people expect it to be one document including all the needed inf...

3 Formulation Development Challenges and How To Face Them
Formulation is a critical step in any product development, however, it is a complex process and formulators face a great amount of challenges when developing a recipe. They have to take into considera...

6 Essential Steps in New Cosmetic Product Development
In the growing industry of personal care and cosmetic, brands and manufacturers need to continuously create and innovate. The collection turnover is getting shorter due to seasonal constraints and fas...

Food Regulations: How to Facilitate Your Product Development in a Global Regulatory Environment
Food regulations are continually evolving with significant differences from one country to another, impacting each and every department of a company. In fact, those regulations impact every step of a...

Which Tests to Ensure Cosmetic Product Safety?
When it comes to cosmetics, product safety is one of the biggest concern of either legal administrations, consumers or manufacturers. Indeed, manufacturers have the responsibility of ensuring that eac...

Making Sense of the Clean Label Concept
Clean Label is there to stay. As said by Lu Ann Williams, Innova Market Insights: “It’s not really a trend anymore, it’s the new rules of the game”. Over the past few years, consumers have been lookin...

Globally Harmonized System: How Does it Affect your Products?
The Globally Harmonized System of Classification and Labeling of Chemicals (GHS) is an international system created by the United Nations in order to standardized classification and labeling of chemic...

Cosmetic Regulations in the USA – All You Need to Know
In the USA, the Food and Drug Administration (FDA) under the Food, Drug, and Cosmetic Act regulate cosmetic products. FDA gives the standards but most of the states have their own regulations such as...

4 Trends in Food and Beverage Formulation
The food and beverage industry has been very dynamic in following trends in order to stay ahead of the game. Taking into consideration those trends, companies largely invest in new product development...

Cosmetic Regulations in California: What You Should Know
Each 50 states of the U.S. are under national and federal regulations. It is not uncommon that states have different regulations from others. This is exactly what happens in the state of California, e...

What to Know About the Nutrition Facts Label Updates
The new nutritional facts label should have been enforced in July 2018. However, the FDA is proposing to extend the compliance deadline. Food manufacturers will have until January 1, 2020 to comply wi...

PLM for Cosmetics: Why Do You Need to Invest?
Cosmetic manufacturers and brands are subject to regulations requirements that are more and more strengthened all over the world. Cosmetic businesses more than ever, must optimize their innovation pro...

Canadian Regulations for Cosmetic Products at a Glance
One of the most important component of new product development concerns the product compliance in the country where it is going to be sold. Indeed, each country has its own regulations especially when...

Sustainable Packaging: Why You Need to Step Up Your Game?
When it comes to packaging, brands have an ocean of choices: color, texture, shape, material, size, etc. And we all know how packaging is a key part of the customer experience and how it is determinan...

Food Fraud – Are You Really Eating What You Think You Are Eating?
Thinking about counterfeit goods, clothing and accessories are usually the first things that come to mind. However, the food and beverage industry is also concerned and fake food scandals have drawn a...

5 Major Trends in the Food and Drink Industry to Follow in 2018!
What are the 5 biggest global trends in the food and drink industry for 2018? Based on an exclusive consumers’ research and experts’ analysis, Mintel has recently launched its 2018 trends study around...

4 Major Packaging Trends to Follow in 2018!
Packaging has become, throughout the years, a significant component for manufacturers when it comes to new product development or renewal. It has evolved in several ways; manufacturers uses it as a wa...

Cosmetic Trends: Lipstick throughout history
Lipstick is an iconic part of the cosmetics industry. Constantly being renewed and innovated, lipstick has changed significantly over the centuries and has also changed in what it represented. (mo...

USDA: The umbrella department of consumer safety
The U.S. Department of Agriculture – USDA is often confused with the Food and Drug Administration –FDA, due to some similar regulations such as product’s traceability and safety. However, there are so...

4 major cosmetic trends to follow in 2018!
Innovation is constant in the Cosmetics industry. Brands have to renovate their products to continue being in line with consumer demands. Consumers are getting more and more attentive to quality, effe...

FDA 101 : All you Need to Know
If you have already sold products in the United States, you must know that the Food and Drug Administration (FDA) regulates all products containing more than one ingredient. FDA is the authority on pr...

How a PLM Software Can Help You Accelerate Product Innovation?
Product life cycles are getting shorter as the world market becomes more and more competitive. Therefore, being early in the development and marketing of a new product can be a serious competitive adv...

10 Global Trends Impacting the Food Industry
When we talk about globalization and politics we often think of the impacts they have on our direct lives, concerning our jobs, education and direct future. But we often forget to think about some oth...

New in Cosmetics: Activated Charcoal
The cosmetics industry is known for its innovative and “thinking outside the box” products. Latest in the cosmetics industry world has been the use of activated charcoal for whiter teeth and clearer s...

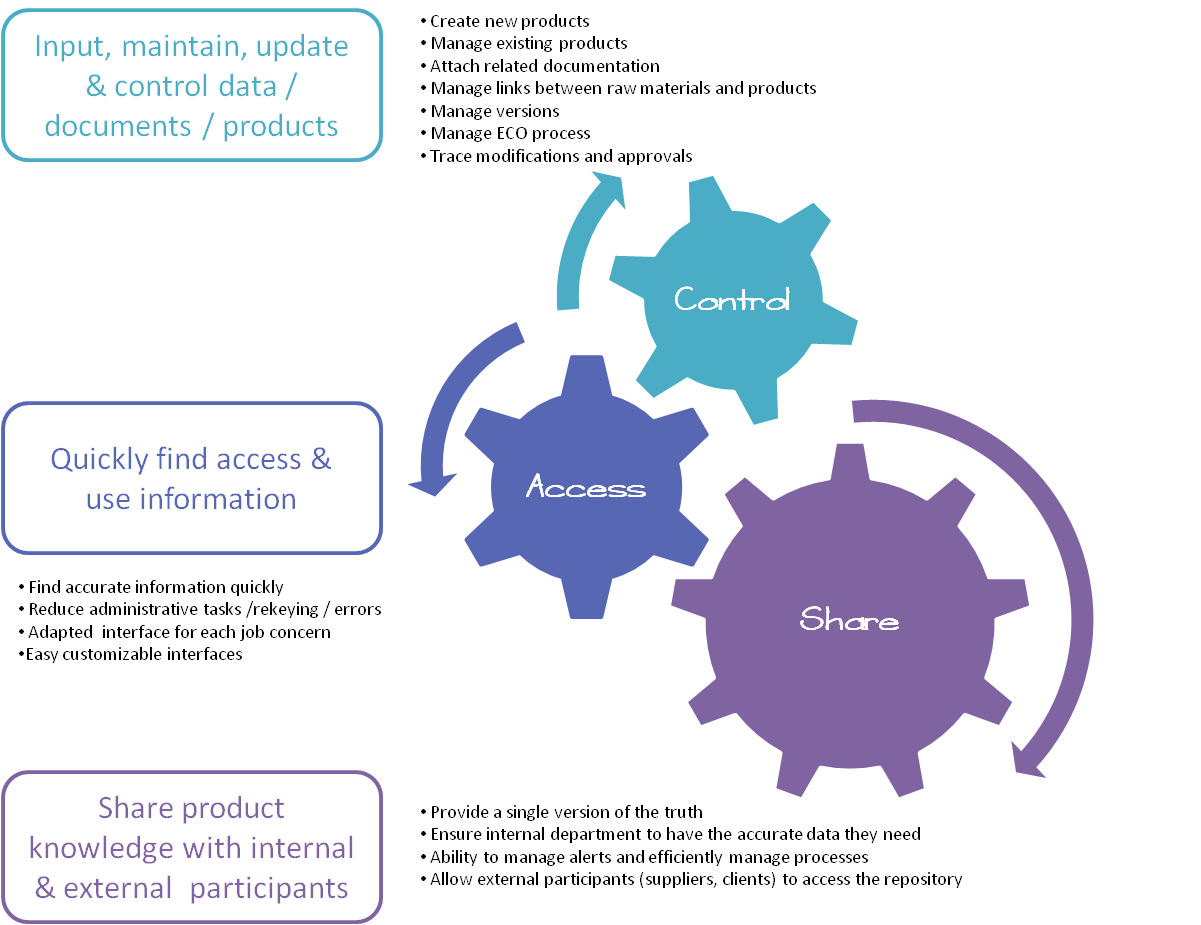
Mismanagement of information: What are the consequences?
Faced with a highly competitive market and increasingly demanding standards, it is becoming essential for companies to be able to effectively manage and track all product-related data, from the design...

Trending Now: Ancient Grains
Although some trends show an increasing drive towards new and evolved products, other trends show the benefits of going back to foods used in the past. According to the National Restaurant Association...

Without silicone, Without sulphates, Without parabens… The “Without” Phenomenon of Cosmetics
Within the last few years, the "without" phenomenon in cosmetics has expanded significantly. Many brands of cosmetics have started openly displaying on the packaging of their products the following cl...

Made In France: Seducing the cosmetics industry abroad
"Made in France" has been associated with quality and luxury for decades. And this is no exception for the cosmetics industry who represents a large amount of France's exports. But what are the hard a...

5 questions when designing product packaging
There are only a number of things a company can change in order to improve its product. But the decision to change the way a package looks is not always a simple one. More likely than not, the design...

Life Cycle of a Cosmetic Product: What you Need to Know
In order to market innovative and high-quality products, the cosmetics industry is subject to increasingly complex regulatory requirements as well as economic and seasonal constraints. To face these n...

What exactly are “SRM”?
The concept of SRM or "Supplier Relationship Management" is being increasingly evoked within companies. In today's day and age, suppliers have an increasingly vital role in the development of new prod...

How to: Manage multiple languages and regulations
The best way to understand the benefits of a PLM is to see a successful example of how they help manage regulations and multiple languages. Take the example of a family business specializing in the...

Avoiding software adoption failures
Choosing the right solution to accompany any development is not as simple as it may seem. Whether are plenty of developments or few, choosing the right solution for the development of that proposal is...

What you need to know about upcycling
Upcycling is the new trend that epitomizes Antoine Lavoisier’s famous quote: "Nothing is lost, nothing is created, everything is transformed" Doing something new with the old has created a real netw...

7 reasons to bet on a PLM solution for cosmetics
Just like with many sectors today, the cosmetics industry is facing major challenges in the face of international regulatory requirements. The best way to overcome these challenges is to continue to i...

How to Design, Develop and Launch new products with speed
Creating and launching products remains one of the main growth strategies for small and large companies. The aim of these companies is obviously to be innovative, attractive and become or maintain the...

Digitalization : what to expect with PLM?
Society is at a point where there is no going backwards. It is all about progress, progress and more progress. Advances in software and technology are allowing for more groups and industries to be int...

Do consumers want green products?
Over the past few years, there has been particular interest paid to the environment and the amount of waste we are releasing into it. However, for most consumers saving the environment doesn’t coincid...

Staying innovative and competitive in the cosmetics industry
It’s a fact. You notice new products every day. And that is simply because of one thing. Innovation. Innovation is the one reason for which people are creating products you haven’t even thought of...

Why are food labels the way they are?
Have you ever wondered why nutrition labels came to have certain pieces of information on them or why they look how they do? Well hopefully this article will answer all your questions. The nutritio...

History of FSMA
At first, food was hunted for but as societies developed people bought from a nearby market and after the industrial revolution and a new form of living people had to buy their food in impersonal supe...

Technical Reference- Steering evolution is necessary
In one of the first articles of the blog, we emphasized the importance of managing the evolution of data and thus having a dynamic vision rather than static information. Since the subject has recently...

PLM: A software package suited to SMEs and major accounts
Naturally, the more the organization of a business is complex, the more the implementation of a PLM may encounter difficulties. However, above all else, the inertia of the company and the resistance t...

How to optimize and better manage your Return On Innovation (ROI)?
Last June we launched a survey to better understand the expectations of the food industry and consumer products on the theme of ROI of innovation and in particular the perception of companies. I invit...

Excel isn’t always the best adapted to your needs
Microsoft excel is one of the most used office software in the corporate world as well as for maintaining contacts books, making calculations, developing dashboards and for using data for charts and...

How to Effectively Manage Customer Complaints
Every business should be listening to its customers in order to react quickly to any problems with their products. A defective product or health or sanitary alerts are significant problems that req...

What are the stakes for the Agro-Food industry in 2030?
The agricultural and Agro-Food industry are sectors that will be present and important in the future due to the population growth and the increase in cost of primary materials -between now and 2030 –...

From the idea to the product
The world of the Agro-Food industry as well as that of consumer goods tends to reduce their range of innovation to the new products chosen by the market and by that of consumer’s demands. This “Market...

What Solution to Accelerate Your Product Development?
Today’s society is characterized by competition, regulation, and demands. With the ever changing economy and internationalization, the pressure on prices continues to increase and this is no different...

Packaging Management: The PLM
Packaging Management: The PLM A Key Solution to optimizing your graphic Chain Sight is one of the most important senses that we possess. And the way things look such as the graphic design of the...

Industrial Food Processing: Avoid Software Adoption Failures
Proposals made to professionals in the food processing industry are plentiful. And yet, choosing the right solution to accompany the proposal's development is not just choosing the solution that ha...

Improve traceability with a PLM solution
Food safety is one of the most significant issues for consumers in developed countries. Producers, manufacturers or retailers must be able to provide accurate and verified document regarding food sour...

Expand product shelf-life and inform consumers with “smart” packaging
In the food industry, packaging has always been a very important part of the product. It's primary function is to contain and protect food therein, ease storage and transportation and also to communic...
Why improving collaboration with suppliers is so important?
To overcome today’s challenges, smart retailers are taking a more focused look at collaborating with their supplier, seeing them as partners. A successful retailer-supplier collaboration can help smoo...

Proactively manage food safety and compliance with PLM
48% of food and beverage manufacturers noted that ensuring compliance with current and future regulations is their top pressure. Legal standards As, mandates such as the Food Safety Modernization Ac...

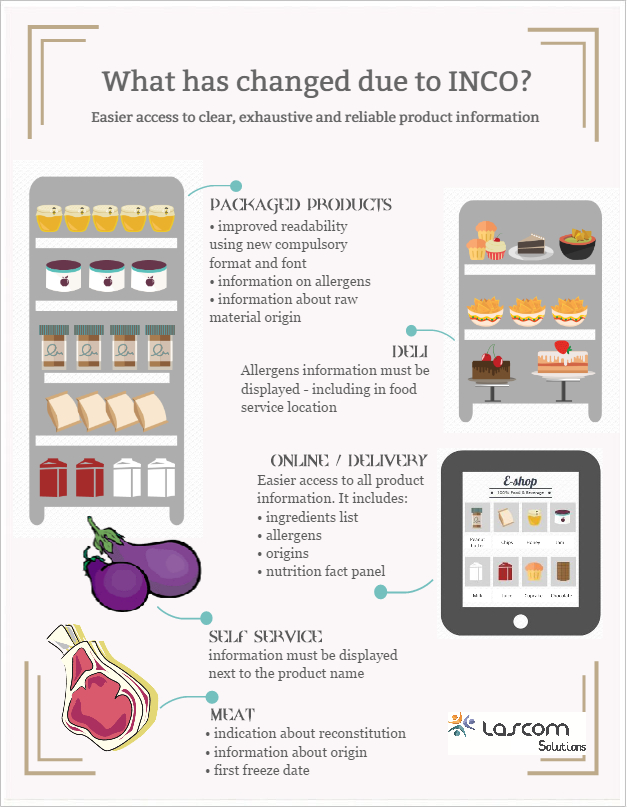
Compulsory changes on-pack / menus due to European 1169/2011 (INCO) regulation
The objective of the regulation is to ensure a high level of consumer protection, to simplify and modernize legislation, and to harmonize the national legislation currently in force. This regulation g...

Hidden sugar and WHO recommendations
A recent WHO survey shows that 16 million people are dying of non genetic disease such as diabetes, cancer or heart disease each year. These diseases are due to a non-healthy lifestyle including high...

Improve Food and Beverage manufacturers and retailers allergen management
More than 200,000 emergency visits are due to Food allergies each year. Up to 15 million Americans suffer from food allergies, affecting 1 in every 13 children under the age of 18 and bringing the eco...

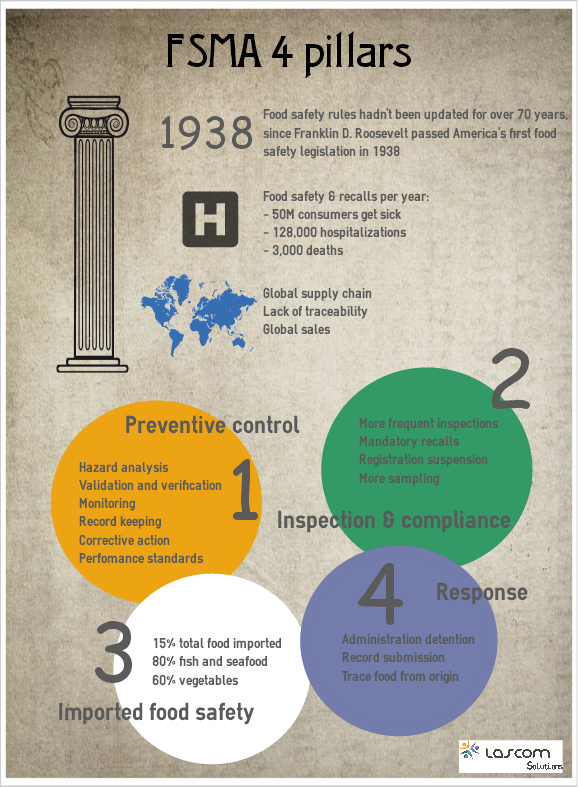
FSMA key pillars
FSMA has a core framework that rests on four key pillars. Preventive Controls Hazard Analysis: You'll need to have plans in place to identify food safety hazards (any property or condition that...
INCO changes
Last December, the new E.U. n°1169/2011 regulation, commonly referred to as INCO (Information to Consumers), was put in place to enhance food labeling. The regulation has already led to significant ch...
FSMA changes
The Food Safety Modernization Act (FSMA) was signed on January 4, 2011. Some requirements are already in place but it globally seems to take more time than expected to implement the whole regulation d...

Key innovation drivers in FMCG
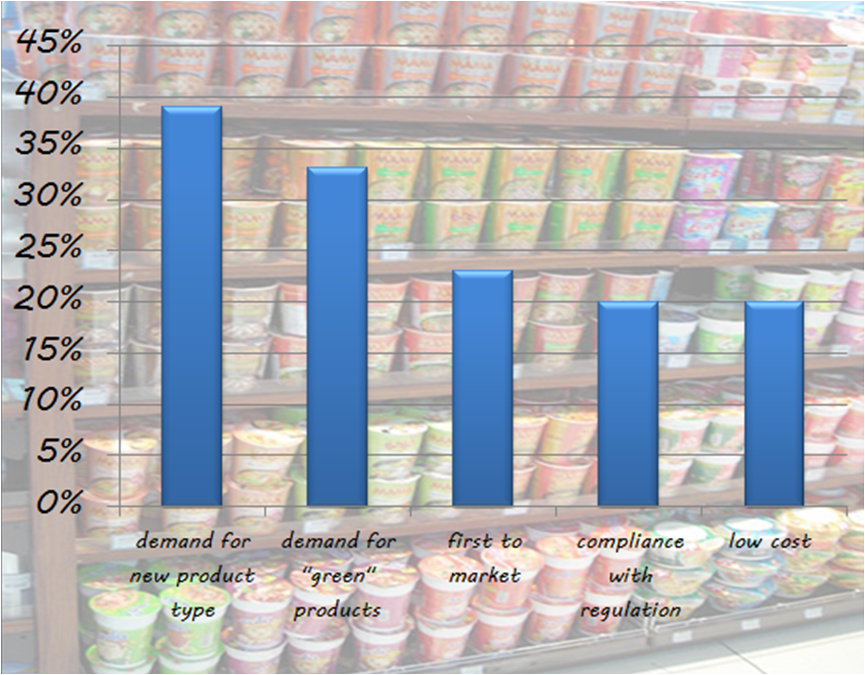
According to an Aberdeen Group survey, the top five pressures driving the FMCG companies are: demand for complete new product type demand for "green" or "clean" products first to market...
Food fraud
NSF International defines food fraud as: "the deliberate substitution, addition, tampering or misrepresentation of food, ingredients or packaging". The 2013 horsemeat scandal and the Elliott report co...

Monin, Leader in Syrups For Professionals, Selects Lascom For Global Product Portfolio Management
Product Lifecycle Management Solution Supports Global Product Management and Development Operations. Lascom today announced that Monin, a global leader in premium syrups, has selected its CPG Solut...

Cosmetics and personal care : how PLM solution assists companies with daily challenges?
To remain competitive, personal care and cosmetics professionals have to be extremely aware of new trends and boost their innovation processes. Reduced time to market, ineffective collaboration, out o...

NPD for the ageing population
In the collective mind, soft drinks are for young and dynamic people. Major brands have always focused on the youth market -10 to 24 year olds- but the beverage industry is changing. Pioneers identify...

New trend: gluten-free products
The demand for gluten-free foods is booming. Some consumers are affected by celiac disease, wheat allergy or are gluten-intolerant or gluten-sensitive, but a large majority of gluten-free products con...

Challenges for PLM in the food industry
The following are few of the questions that professionals in the food industry or food service may ask when they do not have a PLM: How to create a new recipe from an existing one? How to gene...

Improve growth and margins with PLM
PLM (product lifecycle management) is a key element in business success. A study has shown that an effective management tool considerably helps business to not only better manage their product portfol...
Innovation trends in food industry
Innovation is a key factor in the food and beverage sector. Developing a product portfolio by surfing on new consumers' trends can enable companies to achieve improved margins and healthy growth. Quic...

Market innovation: Whole grain trend
According to a 2014 General Mills survey, US people eat 48% more whole grains now than in 2009. It is an impressive rise to compare with the less than 1% growth shown between 2000 and 2004. A whole gr...

Innovation in dairy product : The Greek yogurt star
Consumer buying behavior has changed tremendously in industrialized countries. Food industries try their best to adapt but raw material price increases, tighter regulations, higher energy and labor co...

How to cope with multi-regulations?
It could be extremely difficult to deal with various countries regulations when selling globally. Even if norms and certifications tend to be international (ISO, HACCP, OHSAS...) the majority of rule...

Palm oil: The traceability challenge for food industries
Talking about palm oil is very trendy. A few years ago, Greenpeace opened a new debate with the Kit Kat ads and orangutans protection event to find a solution to the palm oil challenge. At that time c...




























